Where Does Basic3QAQC Fit?
From Rule Based QA/QC to Proactive Quality Management
Most database QA/QC modules are built around rule-based validation. Predefined limits are applied to incoming assay data, and results are flagged when they fall outside those limits. This approach is essential for maintaining control, consistency, and auditability within a data management system.
However, rule-based systems are inherently reactive. They indicate when something has already gone wrong, but they do not explain why it happened or whether it forms part of a broader trend. In practice, this often results in time being spent investigating individual failures without a clear understanding of overall laboratory performance.
Basic3QAQC adds a proactive layer by focusing on how the laboratory is performing, rather than only whether results pass or fail.
By analysing both historical and current data, it becomes possible to identify shifts in precision and accuracy, recognise emerging trends, and detect early signs of deterioration or bias before they develop into larger issues.
Rule based system
“Did this result pass or fail?”
Basic3QAQC
“Is the laboratory performing as expected, and is that performance changing over time?”
Used together, they provide a more complete QA/QC framework, combining control with understanding and allowing QA/QC teams to move from reacting to problems toward actively managing performance.
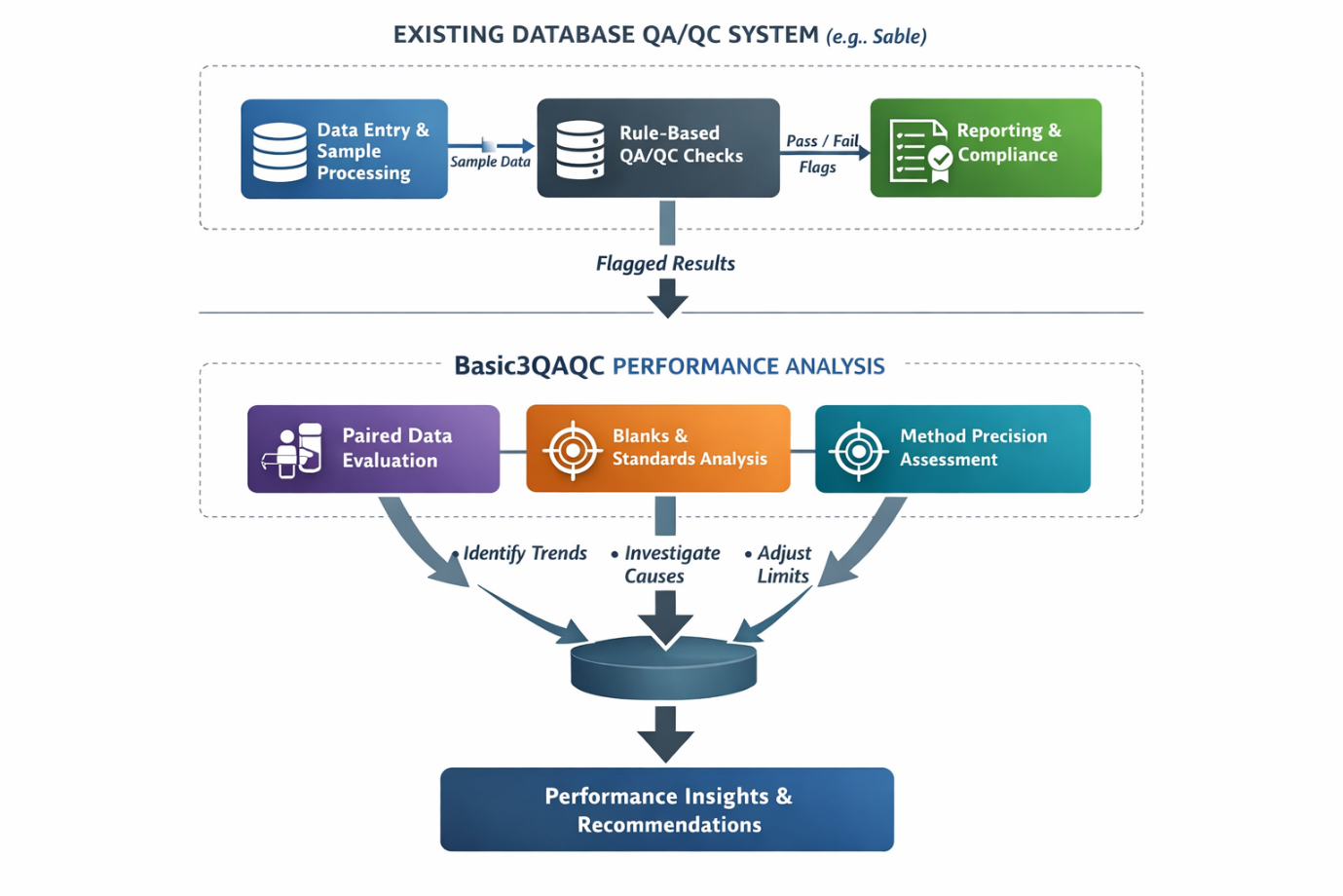
Basic3QAQC — Core Evaluation Tools
Basic3QAQC provides a performance-based evaluation of laboratory data to determine whether assay results are fit for purpose. This is achieved through three core tools:
Paired Data
Precision
Assesses the consistency of laboratory results using duplicates, replicates, and check samples. It quantifies both within-laboratory and between-laboratory precision and highlights changes in variability over time.
Blanks and Standards
Accuracy
Monitors accuracy and contamination by comparing results to Certified Reference Materials and blanks. It identifies bias, drift, and contamination trends across the dataset.
Method Precision
Performance-based limits
Defines expected laboratory performance by calculating confidence intervals based on real data. These limits are used to identify meaningful outliers and ensure QA/QC criteria are aligned with actual laboratory capability.
Practical Outcome
Together, these tools move QA/QC beyond simple pass-or-fail rules by:
- ✓ Quantifying actual laboratory performance
- ✓ Identifying trends and emerging issues early
- ✓ Applying limits that reflect real capability
- ✓ Supporting consistent and defensible QA/QC decisions
In combination with a database QA/QC system, Basic3QAQC provides the analytical layer needed to understand not just whether results fail, but whether the laboratory is performing as expected.
Reporting and Usability
Basic3QAQC is designed to work with defined datasets, allowing QA/QC to be assessed in a structured and controlled way. This makes it easier to review specific programs, laboratories, or reporting periods without unnecessary noise from unrelated data.
A key strength lies in how results are presented. The Blanks and Standards evaluation provides a consolidated summary across all elements in the dataset. This gives a clear, high-level view of laboratory performance in a single output, which can be used directly in internal reporting and significantly reduces reporting time.
At the same time, detailed element-by-element reports are available for further investigation. This allows users to move efficiently from a broad overview to specific technical issues where required. This combination of summary and detail ensures that QA/QC outputs are both practical to use and technically robust.
Method Precision — Practical Value
Method Precision provides a structured, data-driven way to understand and manage laboratory performance based on actual historical results. Instead of relying on fixed or assumed limits, it defines what is realistically achievable for each method, element, and laboratory.
This allows QA/QC teams to move from reactive checks to a more proactive and informed approach.
Key applications include:
Monitoring performance over time
Establishes a baseline from historical data and tracks trends in outliers. A gradual increase in outliers is often the first indication of issues such as increased sample load, instrument drift, or process changes.
Setting realistic QA/QC limits
Defines limits based on actual laboratory capability, reducing unnecessary investigations and ensuring decisions are technically defensible.
Review of CRM performance
Evaluates whether Certified Reference Material limits are appropriate for routine laboratory conditions and highlights where adjustments may be required.
Comparison between laboratories
Provides a consistent, data-driven basis for comparing laboratory performance using precision and confidence intervals.
Defining the noise range
Identifies the concentration range where results remain reliable and where analytical variability begins to dominate.
Recap
Basic3QAQC strengthens QA/QC by aligning quality expectations with actual laboratory performance.
It enables earlier identification of issues, more appropriate limits, and more consistent decision making. When used alongside a database QA/QC system, it ensures that data is not only controlled but properly understood.
See Basic3QAQC in Action
Request a demo, download a trial, or head back to the B3Q Toolkit overview.
